Intuitive Surgical
(NASDAQ: ISRG)
has received approval from regulatory authorities in China to market its Ion surgical robot system.
BTIG analyst Ryan Zimmerman said today that the National Medical Products Administration in China approved the system. This occurred through Intuitive’s partnership with Shanghai Fosun Pharmaceutical, a venture established in 2016.
The major regulatory nod in China follows last week’s FDA approval for the company’s next-generation da Vinci 5 surgical robot.
Intuitive Fosun now produces most of the biopsy needles used in the Ion system and has capacity for about 70% of the global demand, Zimmerman says. Zimmerman and BTIG’s Iseult McMahon said in their report that they expect Intuitive to “ramp up Ion production and sales in China rapidly as a result.”
The analysts said that, along with regulatory approval, Intuitive is launching a large clinical trial in China. It wants to evaluate the diagnostic yield of Ion compared to electromagnetic navigation bronchoscopy in patients undergoing transbronchial sampling procedures for peripheral pulmonary nodules. The company expects to complete the trial in 2026.
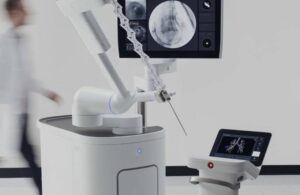
The FDA cleared the Ion system in 2019 as a robotic-assisted, catheter-based platform. Ion enables minimally invasive biopsy deep within the lung. Fiber optic shape sensor technology along the catheter’s length provides its precise location and shape information throughout the procedure.
Zimmerman lists Ion’s largest competitor as the Monarch flexible endoscopic robotic system from Johnson & Johnson MedTech. Monarch picked up a regulatory nod in China for robotic-assisted bronchoscopy in November 2023.