Tandem Diabetes Care (Nasdaq:TNDM) announced positive study data for its artificial pancreas using Control-IQ technology.
The study demonstrated improved blood sugar control in children ages 2-6 with type 1 diabetes. It evaluated an artificial pancreas using the Tandem t:slim x2 insulin pump with Control-IQ technology. Results were published in the New England Journal of Medicine.
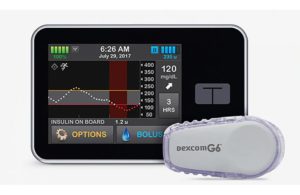
Tandem’s Control-IQ technology automatically monitors and regulates blood glucose. The artificial pancreas utilizes an insulin pump with advanced algorithms based on glucose monitoring from the Dexcom G6 CGM. Then it adjusts the insulin dose as needed. Control-IQ has FDA clearance for people ages six and older with type 1 diabetes.
Get the full story at our sister site, Drug Delivery Business News.