(Image from Lyra Therapeutics)
Lyra Therapeutics (NSDQ:LYRA) announced today that it initiated the Phase 3 Enlighten I trial for its LYR-210 treatment.
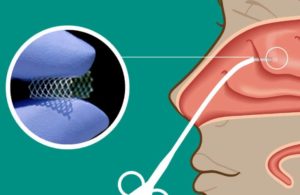
Watertown, Massachusetts-based Lyra’s Phase 3 Enlighten I clinical trial will evaluate LYR-210, delivered through Lyra’s proprietary XTreo drug-eluting implant, in adult, surgically naïve chronic rhinosinusitis (CRS) patients. The company designed LYR-210 for delivery in a brief, non-invasive, in-office procedure with up to six months of continuous anti-inflammatory medication in the sinonasal passages.
Get the full story at our sister site, Drug Delivery Business News.