
You know the big hitters in surgical robotics. Here are some of the companies you may not know about, but you should keep an eye on them.
Last month, MassDevice compiled a list of 16 surgical robotics companies you need to know. That list included the usual suspects, like Intuitive, Medtronic, Johnson & Johnson and more. But, a list of 16 companies hardly scratches the surface.
Competition continues to heat up what is already one of the hottest spaces in medtech. Here are eight more companies that made waves in robot-assisted surgery recently:
| Company | Robotic system |
| CMR Surgical | Versius |
| Medical Microinstruments | Symani |
| Levita Magnetics | MARS |
| Virtuoso Surgical | Virtuoso System |
| eCential Robotics | Surgivisio |
| Globus Medical | ExcelsiusGPS |
| Distalmotion | Dexter |
| Quantum Surgical | Epione |
CMR Surgical
Cambridge, United Kingdom-based CMR Surgical took a massive step forward in 2021.
In June of last year, the surgical robotics developer raised $600 million in a Series D financing round. SoftBank Vision Fund 2 and Ally Bridge Group led the financing.
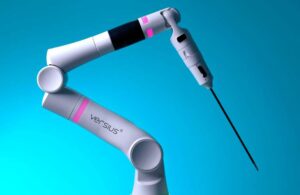
The company’s Versius robotic surgery system is used by surgeons across Europe, the Middle East, Asia, Australia & Latin America. CMR announced the completion of 5,000 surgeries around the world earlier this year.
The company’s website reads: “Versius has been designed to enable surgeons to perform more minimal access surgery, so that more patients can have access to the highest quality of surgical care.”
The platform features freedom of port placement. This best suits the needs of each patient, allowing surgeons to operate the way they did laparoscopically, but with the benefits of robotic surgery, according to the company. It has a small, lightweight and modular design that can be moved “effortlessly.”
Versius’ small-scale design — enabled by v-wrist technology — optimizes it for any virtually any OR, the company says. Its collaborative arms and bedside units can be set up and adjusted to provide direct access to the patient. The system includes a suite of fully wristed instruments and enhanced 3D HD vision.
Altogether, this gives surgeons high levels of accuracy when performing complicated procedural steps or operating in hard-to-reach areas.
Medical Microinstruments
Pisa, Italy–based Medical Microinstruments designed its Symani to address the challenges of microsurgery. It features the NanoWrist instruments for accessing and suturing small, delicate anatomy. This includes veins, arteries, nerves and lymphatic vessels as small as 0.3mm in diameter. It provides motion scaling and tremor reduction to allow precise micro-movements.
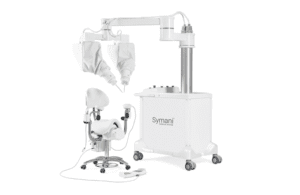
Symani received a CE mark in 2019, and the company intends to accelerate commercialization in the U.S. and Asia-Pacific. It also has plans to advance clinical research through an FDA investigational device exemption (IDE) pivotal study.
The company launched new NanoWrist instruments for Symani earlier this month. The instruments include a new supermicro needle holder and dilator in clinical procedures, both of which produced successful outcomes in Europe.
Medical Microinstruments recently completed a corporate redomicile from Italy to the U.S. Its Center of Excellence facility in Pisa, with 96 employees, will continue to be the hub of its research and development, manufacturing and other business activities. This news accompanied a $75 million Series B financing round this summer.
Levita Magnetics
Levita designed its magnetic-assisted robot surgery (MARS) platform to help surgeons perform more high-volume abdominal procedures using fewer incisions and fewer personnel.

The Menlo Park, California–based company’s system includes less pain, faster recovery and fewer scars. The technology involves a magnet controller outside the body moving tools inside the body. It is built upon the Levita Magnetic Surgical System which received FDA de novo clearance in 2015. That handheld device uses a magnet placed outside of a patient’s abdomen to control a magnetic grasper inside the body during surgery, requiring only one incision instead of two.
Levita’s surgical robotics system improves visualization and helps to maintain surgeon control of instruments while increasing hospital efficiency with fewer assistive personnel required for the procedure. The design specifically targets high-volume ambulatory or same-day discharge abdominal surgeries. Doctors in Chile performed the first procedures with the system last year.
In August, Levita raised $26 million to support the platform.
Virtuoso Surgical
Virtuoso says its technology can provide dexterity in any procedure where tools are delivered through rigid endoscopes. These procedures include urology, gynecology, neurology, interventional pulmonology, orthopedics, thoracic surgery, ear, nose and throat (ENT) and other sub-specialties.

“We are not aware of any technology that accomplishes the dexterity, precision and force of the Virtuoso Surgical device, at such a small diameter,” Robert J. Webster, III, Virtuoso co-founder and president, said in a news release. “It enables surgeons to use two hands to perform more accurate interventions deep in the body through endoscopes.”
The company says its surgical robot system demonstrated feasibility in animal, cadaver and tissue model studies. Surgeries conducted included bladder cancer, uterine fibroids, enlarged prostate, central airway obstruction removal and endoscopic neurosurgery, among others.
Nashville, Tennessee-based Virtuoso’s founders developed the patented technology at Johns Hopkins University and Vanderbilt University. Earlier this month, the company announced a $20 million stock offering.
Virtuoso expects to complete the product design by the end of this year and conduct final testing of the system in 2023.
eCential Robotics
The France-based company picked up a major regulatory win this year with the FDA clearance of its Surgivisio system.

Surgivisio is a robotic 5-axis, optimized 2D x-ray C-arm with integrated 3D image reconstruction and real-time navigation. The eCential system unifies intraoperative 2D/3D imaging, navigation and robotics. The unified system avoids unreliable calibration and registration steps and other pitfalls involved in traditional image-navigation pairing. It also streamlines the surgical workflow and automates technical steps.
The eCential surgical robotics system has a single user interface for all devices. It’s also possible to use the system with any manufacturer’s implants. It currently runs on spine surgery apps. The company plans to expand system uses to other bone surgery indications in the future.
Gières, France–based eCential Robotics plans to start selling the system in North America with help from partnerships with U.S. implant companies. The company has already sold 10 systems in Europe, with more than 2,000 surgeries performed.
Globus Medical

Globus Medical says it designed its surgical robot to enhance safety and improve efficiency for patients, staff and surgeons.
The Excelsius GPS system combines a rigid robotic arm and full navigation capabilities into one adaptable platform for accurate alignment in spine surgery. It aims to enhance accuracy and optimize patient care, the company’s website reads.
In May, Globus announced that it completed its first procedures with the Excelsius3D function. When combined with ExcelsiusGPS, it offers an intraoperative, image-guided robotic navigation solution for improving implant placement accuracy, lowering radiation exposure and shortening operative times. It is now commercially available in the U.S.
Distalmotion
Lausanne, Switzerland–based Distalmotion designed its Dexter system for laparoscopic surgery in general, gynecological and urological surgery.

The company says that the Dexter surgical robotics system combines the affordability of laparoscopy with the benefits of robotic surgery to increase simplicity and versatility in the space. Leading European hospitals are currently spearheading the development of procedural guidelines and training protocols for robotic surgery with Dexter as part of clinical studies and an early adopter program (EAP). Dexter received CE mark approval in Europe in December 2020.
In January, Distalmotion closed a $90 million Series E to support the platform. The company said it intends to use the funding to support the global commercialization Dexter.
Distalmotion said the latest investment arrives as the platform takes its first steps into the operating room, and a commercial rollout awaits.
The company is beginning installments in Western Europe with eyes on global expansion, including in the U.S. The company plans to work closely with the FDA on a U.S. submission supported by the latest financing.
Quantum Surgical
Montpellier, France–based Quantum Surgical designed its robot-assisted Epione system to plan, target, deliver and confirm tumor ablation. The platform is used in minimally invasive liver cancer treatment.

The company said Epione enables clinicians to perform safe and effective percutaneous tumor ablations, expanding the option of such treatment to more patients battling liver cancer. The FDA granted the platform 510(k) clearance in March.
Clearance permits marketing for the Epione interventional oncology robot in the U.S., with Quantum Surgical planning to extend indications for the platform to other organs.
Quantum Surgical says that it also has intentions to develop artificial intelligence (AI)-based decision-support features for the Epione platform.